Noninvasive Hemodynamic Insight for Heart Failure Care
Coredio estimates catheter grade intracardiac hemodynamics using wearables, AI, and digital twins

The Challenge

Insufficient Monitoring
Current standard of care is either non-existent or inaccurate for effective heart failure management

Limited Access Between Visits
Care team dont have access to echocardiograms or catheterizations to get accurate hemodynamic status.

Invasive &
Costly
Accurate insight at-home or out-patient is only available through costly and invasive implantables.
The Challenge

Insufficient Monitoring
Current standard of care is either non-existent or inaccurate for effective heart failure management

Limited Access Between Visits
Care team dont have access to echocardiograms or catheterizations to get accurate hemodynamic status.

Invasive & Costly
Accurate insight at-home or out-patient is only available through costly and invasive implantables.
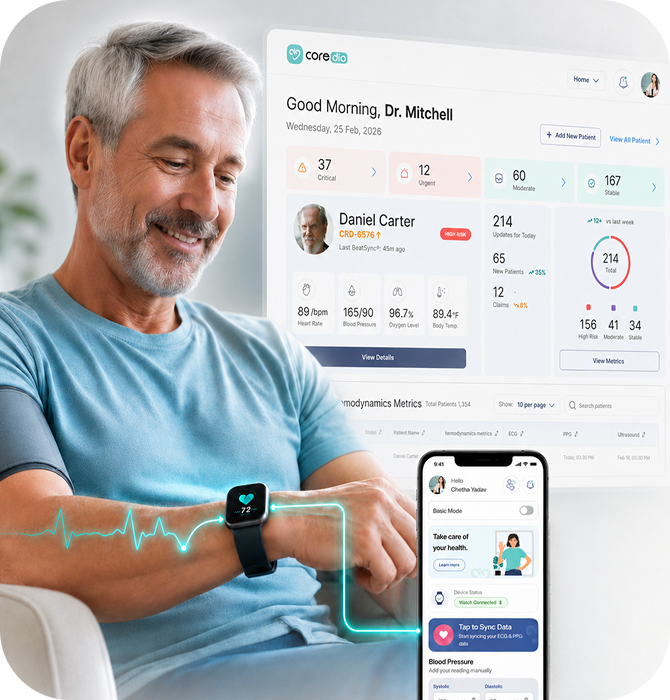
The Platform Solution

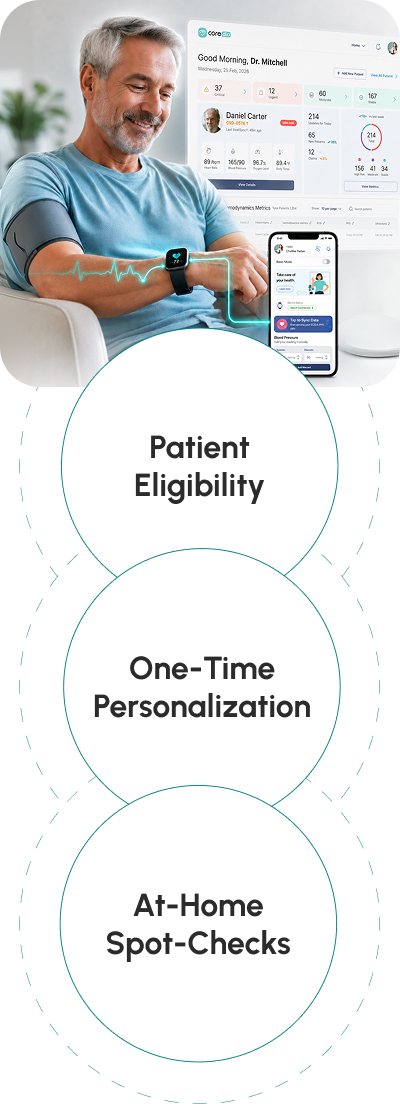
Eligibility


The Solution
 Left Ventricular End-Diastolic Pressure
Left Ventricular End-Diastolic Pressure
Left Ventricular End-Diastolic Pressure: LVEDP reflects left-sided filling pressure and pulmonary congestion. A rising LVEDP can flag worsening heart failure days before symptoms appear.
 Cardiac Index
Cardiac Index
Cardiac Index: Cardiac Index reports cardiac output normalized to body size, indicating whether the heart is delivering enough forward flow to meet the body’s metabolic demand.

 Central Venous Pressure
Central Venous Pressure
Central Venous Pressure: Central Venous Pressure reflects right-sided filling pressure and venous return. Elevated CVP can signal volume overload or right heart strain and is an early indicator of decompensation in heart failure.
 Systemic Vascular Resistance
Systemic Vascular Resistance
Systemic Vascular Resistance: Systemic Vascular Resistance reflects the afterload the left ventricle pumps against. Elevated SVR increases the heart’s workload and can reduce forward cardiac output, often signaling worsening heart failure.


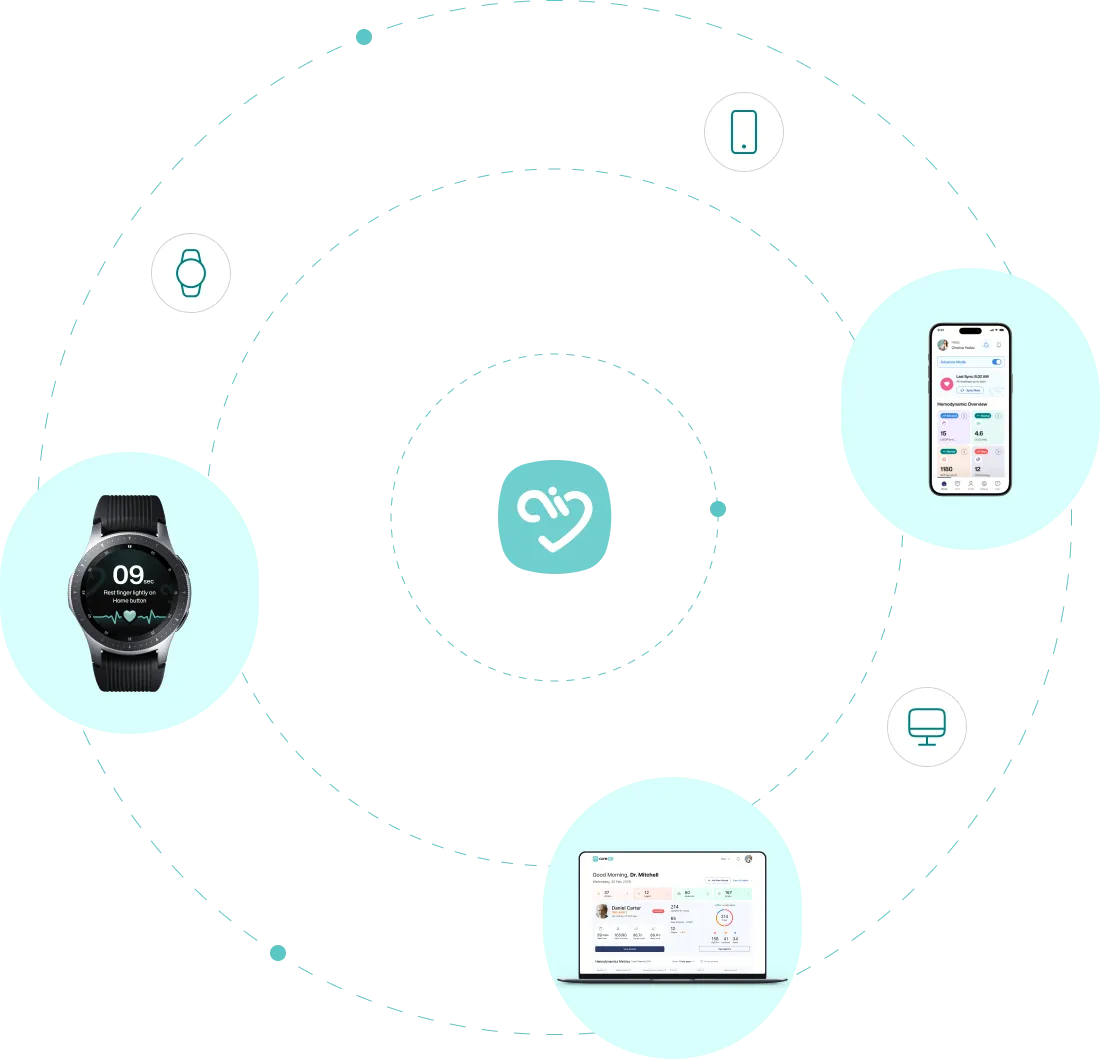
Monitoring Workflow


Patient Personalization
At the initial visit, the care team uses routine, non-invasive checkup tools, while Coredio automatically creates a personalized patient model.
Home Setup
The care team sets up the patient's smartwatch and compatible blood pressure cuff for home use. If the patient does not have a compatible watch, one can be provided.
Remote Monitoring
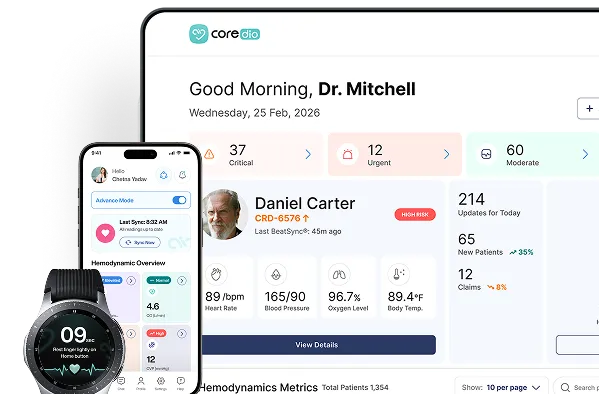
Patients perform daily spot checks at home or in clinic using their smartwatch and blood pressure cuff. Coredio estimates hemodynamic status for clinician review.
Clinical Review & Reporting
Care teams review patientspecific alerts, and intervene early.
-
Patient Personalization
At the initial visit, the care team uses routine, non-invasive checkup tools, while Coredio automatically creates a personalized patient model.
-
Home Setup
The care team sets up the patient's smartwatch and compatible blood pressure cuff for home use. If the patient does not have a compatible watch, one can be provided.
-
Remote Monitoring
Patients perform daily spot checks at home or in clinic using their smartwatch and blood pressure cuff. Coredio estimates hemodynamic status for clinician review.
-
Clinical Review & Reporting
Care teams review patient-specific alerts, and intervene early.
Precision



Testimonial

“Coredio's wearable-derived signals and physics-informed AI could help close the gap between hospital-based measurements and at-home heart failure monitoring, giving clinicians earlier visibility into patient physiology. FDA Breakthrough Device Designation reflects the clinical need this technology addresses.”
“Coredio could bring hemodynamic level insight into the vulnerable post discharge period without invasive devices or extra clinic visits. By using a smartwatch and blood pressure cuff at home, it may help clinicians detect early decompensation and respond sooner.”
“Coredio’s ability to estimate catheterization comparable hemodynamic data from consumer wearables could give clinicians real time visibility between visits. This may support earlier intervention, stronger evidence based decisions, and better outcomes for heart failure patients.”
“Coredio’s digital twin approach may help estimate key hemodynamic metrics from wearable devices, including filling pressures, cardiac index, and vascular resistance. Its FDA Breakthrough Device Designation and TAP Pilot acceptance highlight the promise of non-invasive monitoring.”

Targeted Clinical Outcomes
Validation in progress against right and left heart catheterization at U.S. partner sites.
Does Coredio require me to undergo surgery or implants?
No. Coredio works with a consumer smartwatch and a standard blood pressure cuff. No implants, no needles, no procedures. Coredio’s physics-based AI extracts hemodynamic insights from those signals and surfaces them to your care team.
How does Coredio personalize my care?
Every heart is different. Coredio builds a model from your own baseline measurements, then tracks your individual cardiovascular trends over time and shares them with your clinician. The aim is earlier, smarter intervention before a small change becomes a serious one.
What do I need to get started?
Once Coredio is available through your care team, they will check whether your smartwatch is compatible and provide a standard blood pressure cuff.
Who is Coredio built for?
Both sides of the care relationship. Cardiologists, primary care physicians, Nurses and rest of the care team use Coredio’s dashboard to remotely spot-check heart failure patients and catch concerning trends early. Patients use a smartwatch and our app to stay connected to their care team from home. Caregivers and family members often help with initial setup and patient adherence.
Join the clinicians who are exploring the future of cardiovascular monitoring
with Coredio team
FDA
Disclaimer: Coredio CPSE™ has received FDA Breakthrough Device Designation. The device is not yet cleared by the FDA for commercial use. Indications for Use are subject to FDA review under the 510(k) process.
Disclaimer: Coredio CPSE™ has received FDA Breakthrough Device Designation. The device is not yet cleared by the FDA for commercial use. Indications for Use are subject to FDA review under the 510(k) process.
Join the clinicians who are exploring the future of cardiovascular monitoring
with Coredio team

Disclaimer: Coredio CPSE™ has received FDA Breakthrough Device Designation. The device is not yet cleared by the FDA for commercial use. Indications for Use are subject to FDA review under the 510(k) process.